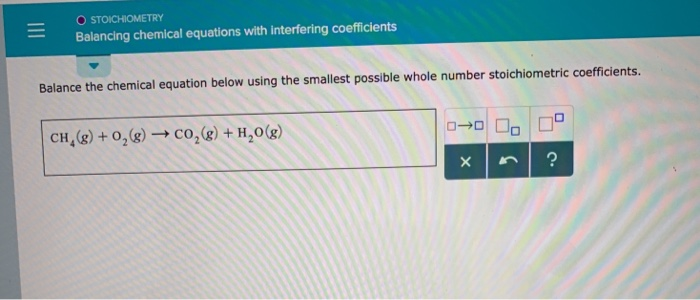
Then check another element, going back and forth from one side of the equation to another, until each element has the same number of atoms on both sides of the arrow. How does one balance a chemical equation, starting with the correct formulas of the reactants and products? Basically, a back-and-forth approach is adopted, counting the number of atoms of one element on one side, checking the number of atoms of that element on the other side, and changing a coefficient if necessary. This online Chemical Equation Balancer Calculator finds the stoichiometric coefficients to balance a given chemical equation, including equations with free electrons and electrically charged molecules (ions) as well as hydrated compounds. the same number of atoms of each element must exist on the reactant side and the product side of the equation. Add a coefficient to the single carbon atom on the right of the equation to balance it with the 3 carbon atoms on the left of the equation. Use a coefficient to balance the single carbon atom. With a user-friendly interface, you can effortlessly input the unbalanced equation, and the calculator performs the tedious calculations in seconds. This means that you will need to balance the carbon atoms first. This sophisticated tool takes the guesswork out of balancing equations. 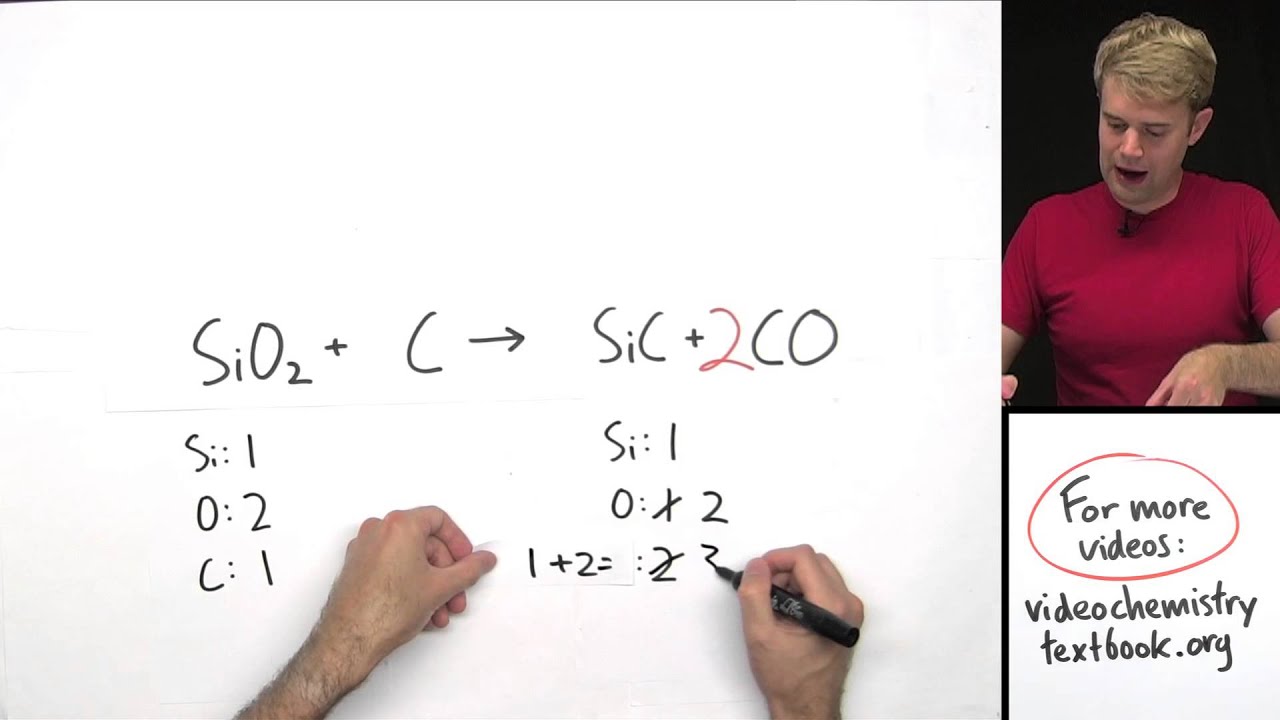
This is important because a chemical equation must obey the law of conservation of mass and the law of constant proportions, i.e. In the digital age, we have a powerful ally at our disposal - the Balancing Chemical Equations Calculator. Write a balanced equation for the reaction of molecular nitrogen (N 2) and oxygen (O 2) to form dinitrogen pentoxide. Example 4.1.1: Balancing Chemical Equations. To balance the number of electrons lost and gained in both parts, multiply both parts with a specific coefficient to get the final equation. O is not balanced the 4 atoms of oxygen on the left does not balance with the 6 oxygen atoms on the right Answer c: Balancing chemical equations involves the addition of stoichiometric coefficients to the reactants and products. These coefficients yield equal numbers of both H and O atoms on the reactant and product sides, and the balanced equation is, therefore: 2H 2O 2H 2 + O 2. The balancing chemical equations calculator highlights you whether your equation is balanced or not.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
